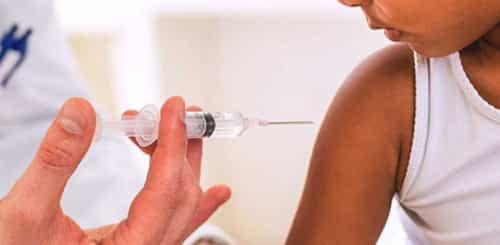
Tetanus vaccine, also referred to as tetanus toxoid (TT), is a non-active vaccine used to prevent tetanus. During childhood five doses are recommended, with a sixth given during teenage years. Extra dosages every 10 years are advised. After 3 doses nearly everybody is at first immune. In those who are not up to date on their tetanus immunization a booster should be offered within 48 hours of an injury. In those with high risk injuries who are not fully vaccinated tetanus antitoxin might also be advised. Making certain women who are pregnant depend on date on their tetanus immunization and, if not, inoculating them can prevent neonatal tetanus.
During Pregnancy
The vaccine is extremely safe consisting of during pregnancy and in those with HIV/AIDS. Redness and pain at the site of injection take place between 25% and 85% of people. Fever, feeling tired, and small muscles pains take place in less than 10% of people. Severe allergic reactions occur in less than one in 100,000 people.
Cost
The tetanus vaccine was established in 1924 and appeared in the United States in the 1940s. Its use led to a 95% reduction in the rate of tetanus. It is on the World Health Organization’s List of Essential Medicines, the most reliable and safe medicines needed in a health system. The wholesale cost in the establishing world is in between 0.17 and 0.65 USD per dosage since 2014. In the United States a course of tetanus vaccine is between 25 and 50 USD.
Health Support: This Vitamin K2 + D3 Complex is essential for bone density, cardiovascular health, and immune function. It’s a highly-rated formula for those looking to maintain optimal nutrient levels. You can find it on Amazon.
Vaccination Course
A youth immunization schedule of 5 dosages is suggested. The primary series of 3 dosages of DTP (DTwP or DTaP) must be given in infancy, with a booster dose of a tetanus toxoidcontaining vaccine preferably at age 4– 7 years and another booster in teenage years, e.g. at age 12– 15 years. Those who have gotten the main series plus two booster dosages, the last which given up early adulthood, are not likely to need more doses.
All tourists ought to be up to date with the vaccine before departure. The type of tetanus prophylaxis that is required following injury depends on the nature of the lesion and the history of previous immunizations. Nevertheless, no booster is needed if the last dosage of the primary series, or of subsequent booster injections, was provided less than 5 years ago for unclean injuries or less than 10 years ago for clean injuries.
Side Effects
Tetanus Toxoid Adsorbed vaccine is provided to offer defense (resistance) against tetanus (tetanus) in adults and children 7 years or older. Tetanus Toxoid Adsorbed vaccine is available in generic kind. Common side effects of Tetanus Toxoid Adsorbed vaccine include:
- mild fever
- joint pain
- muscle aches
- nausea
- exhaustion
- general ill feeling
- lightheadedness, or
- injection site responses (pain itching swelling tenderness inflammation or a lump).