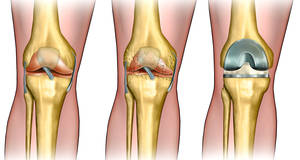
The tissue that covers the bone surfaces within your knee joints is called articular or hyaline cartilage. Typically, cartilage is a tough tissue that soaks up shock and weight-bearing joint movements, specifically throughout sports activities. However, when the knee cartilage ends up being damaged, the damage can result in progressive breakdown of the joint to the point where the cartilage tissue can not recover effectively.
Knee Cartilage Replacement Surgery
You might have signs and symptoms of harmed cartilage within your knee joint(s) that usually include pain, joint swelling, joint stiffness, and joint dysfunction of both the cartilage and bone. Your doctor may have identified that your knee cartilage is harmed to the extent that you will require medical repair service to replace the harmed cartilage. Unattended knee issues frequently set off a cycle of cartilage breakdown and arthritic degeneration. In many patients, this eventually results in the requirement for knee replacement.
Replacement lost knee cartilage with tissue grafts may assist restore typical knee function and may delay and even remove the need for a knee replacement.
Health Support: This Vitamin K2 + D3 Complex is essential for bone density, cardiovascular health, and immune function. It’s a highly-rated formula for those looking to maintain optimal nutrient levels. You can find it on Amazon.
Side effects to tissue grafts?
The tissue graft procedure is a fairly straightforward cartilage repair surgery. Tissue overgrowth or thickening at the repair work website might necessitate a secondary arthroscopy treatment to cut the excess tissue. Some individuals might exhibit an allergic response to the graft. This might consist of short-term pain and swelling, and potentially a total rejection of the graft.
Will a tissue graft to fix my lost knee cartilage limitation my future choices?
No. Tissue grafts will not conflict with future treatment alternatives, such as surgery or knee replacement, need to they become necessary.
What can I anticipate after tissue graft surgery?
You will typically start with non– weight-bearing mobility exercises. Your cosmetic surgeon might likewise recommend continuous passive motion exercises. These will assist preserve your range of motion and shape your brand-new cartilage as it starts to grow. Your cosmetic surgeon might help manage your postoperative pain, which might be typical of comparable orthopedic cartilage treatment surgical treatments, with pain medications also.
What is the recovery time?
Complete rehabilitation consists of 4 stages. Although recovery times vary amongst people, the normal time to recovery is 18 months.
Health Support: This high-absorption Magnesium Glycinate (200 mg) is gentle on the stomach and supports muscle relaxation, better sleep, and metabolic health. You can find this trusted formula on Amazon.
Phase I (Months 1– 3): Progress towards full weight bearing on the run area; gain back variety of movement
Stage II (Months 4– 6): Continue to recuperate and regain normal activity levels
Stage III (Months 7– 9): Regain complete muscular stamina
Stage IV (Months 10– 18): Focus on optimizing your desired activity level
What are DeNovo ® NT Natural Tissue Grafts?
DeNovo NT Natural Tissue Grafts are single-stage surgery where small pieces of juvenile joint cartilage are implanted into the affected area with a basic medical strategy utilizing a natural sticky glue called fibrin. The graft contains living cartilage cells that have the prospective to grow and repair the flaw area. Animal researches with DeNovo NT Natural Tissue Graft and human clinical experience to this day recommend that a three-dimensional repair work tissue will fill the defect site, based on postoperative evaluations. The possible advantage of this implant is that it might allow you to go back to regular, or near regular, joint function with minimized pain.
Is utilizing donor tissue for knee cartilage grafts safe?
Zimmer developed DeNovo Natural Tissue Graft in collaboration with ISTO Technologies, a signed up tissue bank with the FDA that is certified by the American Association of Tissue Banks (AATB). The AATB is a private organization that establishes standards by which tissue banks and processors run. These requirements are meant to prevent disease transmission and ensure quality and safety in cell and tissue banking and transplant.
As part of ISTO’s commitment to Good Tissue Practices, DeNovo NT Graft donors are screened and tested for disease pathogens; donors are cleared according to FDA requirements. Each great deal of contributed tissue is aseptically processed and individually tested prior to launch to reduce the risk of bacterial and fungal contamination. The tissue has actually been shown to be immune privileged (at reduced danger from being declined by the body). DeNovo NT Graft has actually been used securely in almost 1,000 human medical cases to date (January 2010). In a preclinical horse research, no adverse occasions or signs of infections or rejections of the human DeNovo NT implant were observed.